KEY DISTINGUISHING FEATURES OF THE CITRANVI GLOBAL HERPESVIRUS VACCINES TECHNOLOGY PLATFORM DESIGN.
Scientists at the USUHS (Dept of Defense) have been able to genetically construct and purify the hCMV and EBV vaccine candidates in structural-forms and formulations that enhance their ability to produce robust functional antibody responses. These functional antibodies play a key role in:
Scientists at the USUHS (Dept of Defense) have been able to genetically construct and purify the hCMV and EBV vaccine candidates in structural-forms and formulations that enhance their ability to produce robust functional antibody responses. These functional antibodies play a key role in:
- hCMV: Inhibition of viral infection of both epithelial cells and fibroblast cells
- EBV: Inhibition of viral infection of both B-cells and epithelial cells
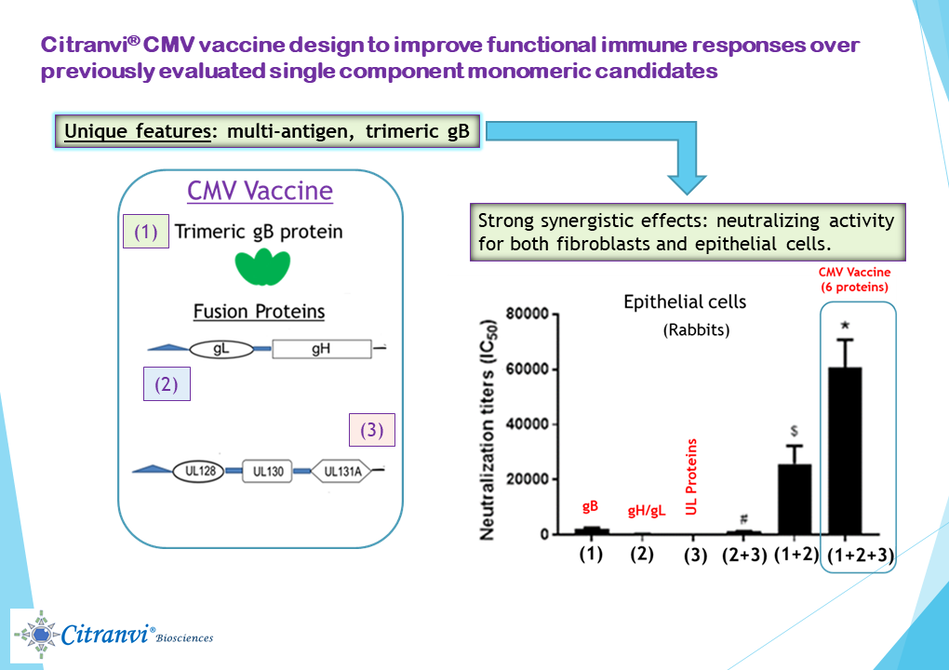
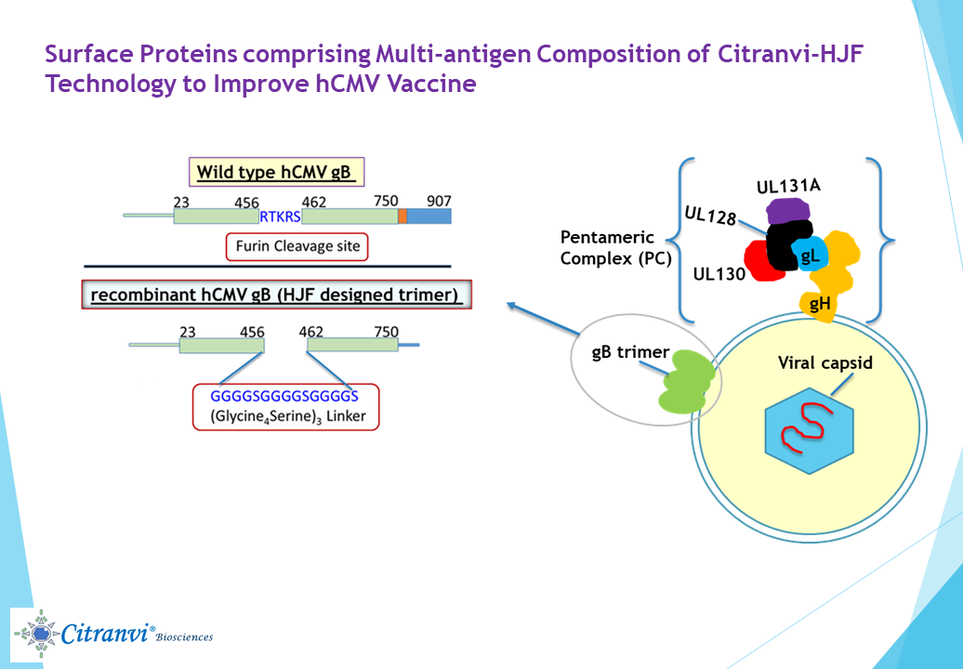
1. CITRANVI GLOBAL MULTIANTIGEN CMV VACCINE: The gB protein on the surface of hCMV virus is important for the fusion of the virus with all the cell types that the virus can infect. Thus, the gB protein was once the leading vaccine candidate for a hCMV vaccine, as a single component. However, the gB protein is present naturally as a trimer on the surface of the virus and the optimal vaccine presentation of the gB protein, therefore, should be as a trimer and not as a monomer.
Structure of hCMV gB protein (trimer)
In addition, now it is known that besides trimeric gB other viral surface proteins are also important for hCMV infection. To improve upon the clinical results obtained earlier with the monomeric form of gB, Citranvi is developing a 6-antigen hCMV vaccine formulation with the protein components. The five-protein antigen components comprising gH, gL together with the UL128, UL130 and UL131A form the pentameric complex (PC). The PC combined with the immunogenic trimeric form of gB protein should provide robust protection from hCMV infections.
A key aspect of the Citranvi Global Multiantigen CMV Vaccine design is the genetic construction of the (a) trimeric gB protein; (b) the genetic fusion protein of gH/gL and (c) the fusion approach involving the three UL proteins for development of a new multi-antigen hCMV vaccine. Immunization of small animals with these purified proteins with a CpG oligonucleotide + aluminum hydroxide adjuvant has shown the following:
(1) Trimeric gB protein elicited higher antibody titers of hCMV neutralizing activity, across different clinical strains, relative to monomeric gB protein.
(2) Immunization with the combination of trimeric gB and gH/gL protein antigens showed strong synergistic effects in elicitation of hCMV neutralizing activity for both infection of fibroblasts and epithelial cells.
(3) Immunization with the combination of trimeric gB, gH/gL and UL protein antigens demonstrated the strongest synergistic effects in elicitation of hCMV neutralizing activity for infection of epithelial cells.
(4) Immunization of rabbits with the combined vaccine mixture of trimeric gB, gH/gL and UL128-UL130-UL131A fusion protein with a CpG oligonucleotide + hydroxide adjuvant showed no antigenic interference in the ability to stimulate antibody response to each protein.
(1) Trimeric gB protein elicited higher antibody titers of hCMV neutralizing activity, across different clinical strains, relative to monomeric gB protein.
(2) Immunization with the combination of trimeric gB and gH/gL protein antigens showed strong synergistic effects in elicitation of hCMV neutralizing activity for both infection of fibroblasts and epithelial cells.
(3) Immunization with the combination of trimeric gB, gH/gL and UL protein antigens demonstrated the strongest synergistic effects in elicitation of hCMV neutralizing activity for infection of epithelial cells.
(4) Immunization of rabbits with the combined vaccine mixture of trimeric gB, gH/gL and UL128-UL130-UL131A fusion protein with a CpG oligonucleotide + hydroxide adjuvant showed no antigenic interference in the ability to stimulate antibody response to each protein.
2. CITRANVI GLOBAL MULTIANTIGEN EBV VACCINE
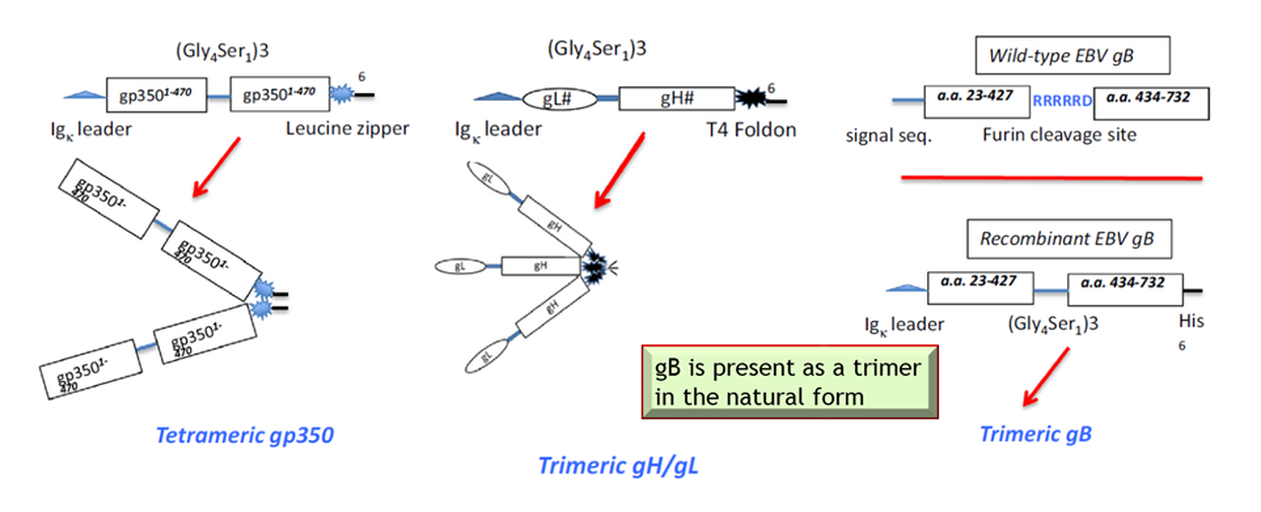
Recent data has demonstrated that in addition to gp350 other viral envelope proteins also play a role in causing EBV infection. For example, the gH/gL and gB proteins coordinately mediate virus fusion and entry into B cells and epithelial cells whereas gp350 mediates attachment to B-cells but not epithelial cells. Important for the Citranvi vaccine design, a more immunogenic, tetrameric form of gp350 protein, in addition to trimeric forms of gH/gL and gB proteins were produced as a new multi-antigen EBV vaccine candidate by scientists at the USUHS.
The Citranvi Global 4-Antigen EBV Vaccine formulation comprising multimeric forms of virus surface proteins gp350, gB and gH/gL has been designed to elicit antibodies that will prevent EBV infection.
Recent data has demonstrated that in addition to gp350 other viral envelope proteins also play a role in causing EBV infection. For example, the gH/gL and gB proteins coordinately mediate virus fusion and entry into B cells and epithelial cells whereas gp350 mediates attachment to B-cells but not epithelial cells. Important for the Citranvi vaccine design, a more immunogenic, tetrameric form of gp350 protein, in addition to trimeric forms of gH/gL and gB proteins were produced as a new multi-antigen EBV vaccine candidate by scientists at the USUHS.
The Citranvi Global 4-Antigen EBV Vaccine formulation comprising multimeric forms of virus surface proteins gp350, gB and gH/gL has been designed to elicit antibodies that will prevent EBV infection.
The novel and proprietary Citranvi global design represents a marked improvement over a monomeric gp350 vaccine candidate from the previous clinical trial that showed 78% efficacy against infectious mononucleosis caused by EBV but did not prevent asymptomatic EBV infection. The pre-clinical data strongly supports that a Citranvi-HJF 4-antigen vaccine formulation, composed of trimeric gH/gL and trimeric gB, along with tetrameric gp350, can prevent EBV infection as well as transmission and would be a major improvement over the clinical results obtained with the monomeric gp350 vaccine.
Using small animal models, immunization studies with these purified proteins with a CpG oligonucleotide plus aluminum hydroxide adjuvant have shown that the multimeric EBV protein antigens induce significantly higher serum IgG titers relative to their monomeric protein counterparts.
The vaccine formulation comprising multimeric forms of gp350, gB and gH/gL proteins should elicit antibodies that will prevent EBV infection in both B cells and epithelial cells, which represents a marked improvement over the monomeric gp350 vaccine candidate from a previous clinical trial. In addition, the higher IgG antibody titers may also enhance protective non-neutralization anti-viral effector functions and mucosal IgG. These data strongly support that a Citranvi Global 4-Antigen EBV Vaccine formulation composed of trimeric gH/gL and trimeric gB, along with tetrameric gp350, can prevent EBV infection as well as transmission.
Using small animal models, immunization studies with these purified proteins with a CpG oligonucleotide plus aluminum hydroxide adjuvant have shown that the multimeric EBV protein antigens induce significantly higher serum IgG titers relative to their monomeric protein counterparts.
The vaccine formulation comprising multimeric forms of gp350, gB and gH/gL proteins should elicit antibodies that will prevent EBV infection in both B cells and epithelial cells, which represents a marked improvement over the monomeric gp350 vaccine candidate from a previous clinical trial. In addition, the higher IgG antibody titers may also enhance protective non-neutralization anti-viral effector functions and mucosal IgG. These data strongly support that a Citranvi Global 4-Antigen EBV Vaccine formulation composed of trimeric gH/gL and trimeric gB, along with tetrameric gp350, can prevent EBV infection as well as transmission.
Extension of Citranvi Global Herpesvirus Vaccine Platform Technology to Other Herpesvirus Pathogens
A significant advantage of the Citranvi herpesvirus vaccines platform is that this technology could be broadly applied to develop additional vaccine candidates against other herpesvirus pathogens such as Herpes Simplex Virus (HSV) and Varicella Zoster Virus (VZV, Shingles). HSV-1 and HSV-2 are alpha herpesviruses for which there are currently no commercially licensed vaccines. A prophylactic HSV vaccine is a WHO-supported global public health priority. HSV causes oral and genital ulcerative lesions, ocular infection causing blindness, encephalitis, meningitis, and neonatal infection.
A significant advantage of the Citranvi herpesvirus vaccines platform is that this technology could be broadly applied to develop additional vaccine candidates against other herpesvirus pathogens such as Herpes Simplex Virus (HSV) and Varicella Zoster Virus (VZV, Shingles). HSV-1 and HSV-2 are alpha herpesviruses for which there are currently no commercially licensed vaccines. A prophylactic HSV vaccine is a WHO-supported global public health priority. HSV causes oral and genital ulcerative lesions, ocular infection causing blindness, encephalitis, meningitis, and neonatal infection.